PHARMACOVIGILANCE UNBOXED
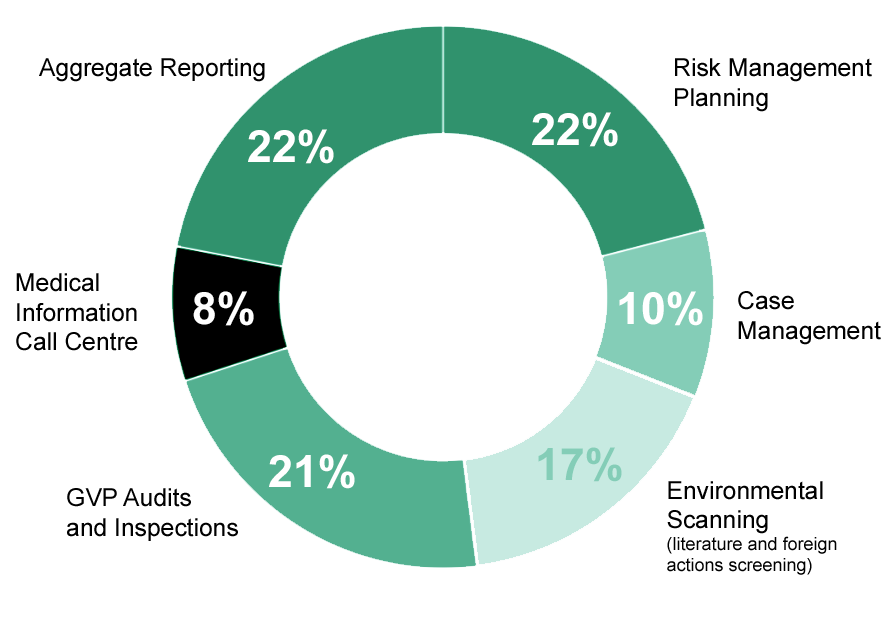
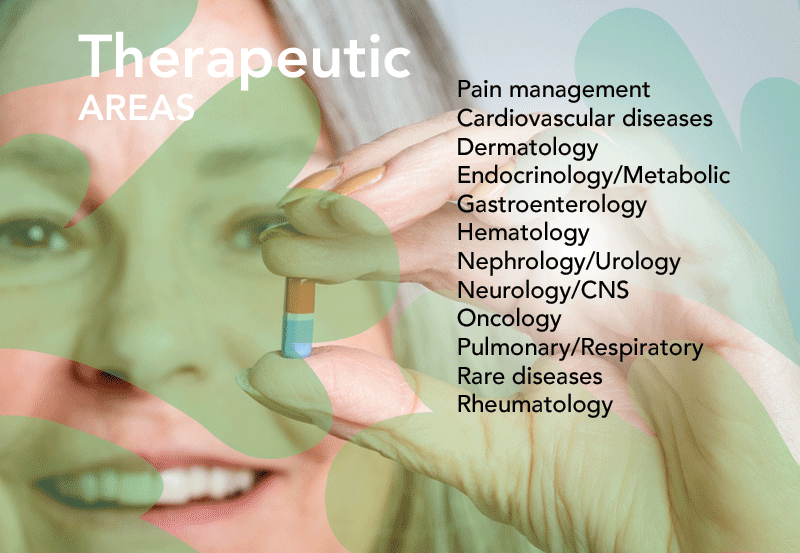
Full suite of Pharmacovigilance (PV) and Medical Information (MI) services delivered by a dedicated team of experienced healthcare professionals. From routine activities to the most complex regulatory challenges, our team provides tailored solutions to navigate the evolving PV landscape with confidence.
We bring clarity to complex processes, ensure compliance with regulatory requirements, demonstrate adaptability in an ever-changing environment, and apply deep expertise to deliver safe, effective, and compliant outcomes for our Partners. Serving Clients of all sizes - from global pharmaceutical leaders to mid-sized innovators and emerging startups - we adapt our expertise to deliver tailored solutions.